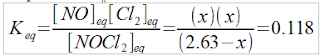I'm working on the hand in right now and I'm just a little stuck on the % Fe question. I have all of the molarities calculated out. Do I simply take the molarity of the samples and divide them by .100 L for the moles and then go to grams to percent? My percentages just seem so small. I'm getting numbers such as .22% Fe. Or do I use the .050 L instead of .100.
If you break this calculation down into its parts, it's not quite as intimidating as it might seem.
Step 1 - Using your Beer's Law plots, calculate the iron concentration in the orange solutions you made in the second week of lab.
Step 2 - How did you make those orange solutions? You should have made them from 1.00mL of the yellow iron solution you made by dissolving your iron ore sample (red powder) in acid. You can use C1V1=C2V2 to determine the concentration of iron in the yellow solution.
Step 3 - Once you know the concentration (moles of iron per liter of solution) of the yellow iron solution, you can calculate how many moles of iron are present in that solution. You made 100.0mL of the yellow solution from your red iron ore sample.
Step 4 - If you know how many moles of iron were present in the yellow iron solution, you know how many moles of iron were present in the red iron ore powder. {Hint: they just might be the same...}
Step 5 - You know how many moles of iron were present in the red iron ore sample, using the Periodic Table you can calculate how many grams of iron were in the red iron ore sample.
Step 6 - If you know how many grams of iron were present in the red iron ore sample, and you know the total mass of the red iron or sample, then you should be able to calculate the percent (by mass) of iron that was present in the red iron ore sample.
For the iron ore samples you all analyzed, the percent iron could have been as low as 9-11% or as high as 33-35%. If you get something lower than 9% or higher than 35%, double check your calculations.